Clinical Profile

Well-tolerated therapy that helps manage the skin manifestations of CTCL*
-
Adverse events (AEs) in clinical trials were primarily related to hypotension secondary to changes in extracorporeal volume (>1%)1
-
In Trial 31:
-
Six serious cardiovascular AEs (5 unrelated to photopheresis) were reported in 5 patients (10%)
-
Six infections were also reported in 5 patients
-
Two were Hickman catheter infections in 1 patient
-
-
Adverse reactions reported from more than 30 years of postmarketing experience include nausea, dysgeusia, photosensitivity reaction, pyrexia, and hypersensitivity reactions, including anaphylaxis and rash1
* UVADEX is indicated for extracorporeal administration with the THERAKOS® CELLEX® Photopheresis System in the palliative treatment of the skin manifestations of CTCL that is unresponsive to other forms of treatment.
THERAKOS Photopheresis Immunomodulatory Therapy Delivers a Meaningful Response
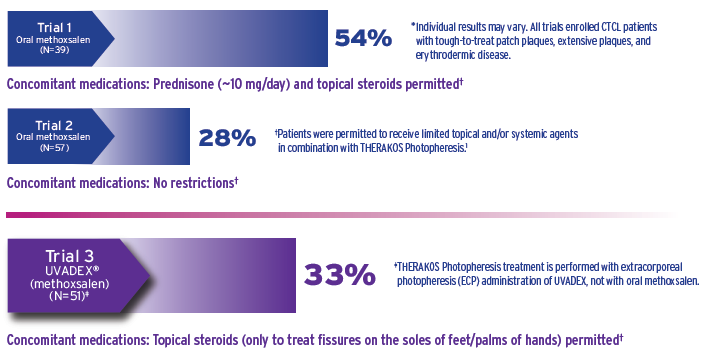
Clinical trial efficacy was established in patients unresponsive to skin-directed CTCL treatments, including tough-to-treat disease1*
-
3 multicenter, single-arm, open-label trials studied efficacy and safety. Success was predefined as a skin score reduction of ≥25% from baseline maintained for 4 consecutive weeks1
-
Across the 3 trials, 147 patients participated, including patients diagnosed with mycosis fungoides and Sézary syndrome, both subtypes of CTCL1,2
-
Although the response rates with UVADEX in Trial 3 and oral methoxsalen in Trial 2 were similar, the possibility that UVADEX is inferior in efficacy to oral methoxsalen cannot be excluded due to the design and size of the clinical trials1
-
In Trial 3, 15 of 17 responses were observed within 6 months and 2 responses were observed after 6 months1
-
The higher response rate with oral methoxsalen in Trial 1 may be partly due to the coadministration of systemic steroids and patients receiving more extracorporeal photopheresis treatments (mean number of treatments: Trial 1=64; Trial 2=31; Trial 3=20)1
-
No patients with disease in the tumor phase were treated and there are no data available regarding efficacy of UVADEX in patients with disease in the tumor phase1
Real-world Experience for Over 30 Years and More Than 1 Million Treatments Administered3
Hear from patients with CTCL skin symptoms treated with THERAKOS Photopheresis

Actual patient who has been
compensated for sharing her photopheresis experience.
Patient profile: Ann
The most obvious initial improvement that I experienced was relief from persistent itchiness. This may not seem like an important improvement to people who have not experienced the itching and other skin issues caused by CTCL.

Actual patient who has been
compensated for sharing his photopheresis experience.
Patient profile: Lou
When I went on vacation, I missed a treatment, and I didn’t go for 2 months. Big mistake. I started flaring up. My skin became red and hot, and that chapped-and-chafed look came back. That’s how I really knew that photopheresis was having an effect.
References:
-
UVADEX (methoxsalen) Sterile Solution (prescribing information). Therakos Inc; March 2023.
-
Data on File — Ref-VV-05182. Mallinckrodt Pharmaceuticals.
-
Data on File — Ref-VV-02008. Mallinckrodt Pharmaceuticals.
INDICATIONS AND USAGE
UVADEX® (methoxsalen) Sterile Solution is indicated for extracorporeal administration with the THERAKOS® CELLEX® Photopheresis System in the palliative treatment of the skin manifestations of Cutaneous T-Cell Lymphoma (CTCL) that is unresponsive to other forms of treatment.
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
UVADEX is contraindicated in:
-
Patients exhibiting idiosyncratic or hypersensitivity reactions to methoxsalen, other psoralen compounds, or any of the excipients
-
Patients possessing a specific history of a light-sensitive disease state, including lupus erythematosus, porphyria cutanea tarda, erythropoietic protoporphyria, variegate porphyria, xeroderma pigmentosum, and albinism
-
Patients with aphakia because of significantly increased risk of retinal damage
-
Patients that have contraindications to the photopheresis procedure
WARNINGS AND PRECAUTIONS
-
Patients who are receiving concomitant therapy (either topically or systemically) with known photosensitizing agents such as anthralin, coal tar or coal tar derivatives, griseofulvin, phenothiazines, nalidixic acid, halogenated salicylanilides (bacteriostatic soaps), sulfonamides, tetracyclines, thiazides, and certain organic staining dyes such as methylene blue, toluidine blue, rose bengal, and methyl orange may be at greater risk for photosensitivity reactions with UVADEX
-
Oral administration of methoxsalen followed by cutaneous UVA exposure (PUVA therapy) is carcinogenic. Methoxsalen also causes DNA damage, interstrand cross-links and errors in DNA repair
-
Methoxsalen may cause fetal harm when given to a pregnant woman. Women of childbearing potential should be advised to avoid becoming pregnant. If UVADEX is used during pregnancy, or if the patient becomes pregnant while receiving UVADEX, the patient should be apprised of the potential hazard to the fetus
-
Severe photosensitivity can occur in patients treated with UVADEX. Advise patients to wear UVA absorbing, wrap-around sunglasses and cover exposed skin or use a sunblock (SPF 15 or higher), and avoid all exposure to sunlight for twenty-four (24) hours following photopheresis treatment
-
After methoxsalen administration, exposure to sunlight and/or ultraviolet radiation may result in "premature aging" of the skin
-
Since oral psoralens may increase the risk of skin cancers, monitor closely those patients who exhibit multiple basal cell carcinomas or who have a history of basal cell carcinomas
-
Serious burns from either UVA or sunlight (even through window glass) can result if the recommended dosage of methoxsalen is exceeded or precautions are not followed
-
Exposure to large doses of UVA light causes cataracts in animals. Oral methoxsalen exacerbates this toxicity
-
Safety in children has not been established
-
Thromboembolic events, such as pulmonary embolism and deep vein thrombosis, have been reported with UVADEX administration through photopheresis systems for treatment of patients with graft-versus-host disease, a disease for which UVADEX is not approved
ADVERSE REACTIONS
-
Side effects of photopheresis (UVADEX used with the THERAKOS Photopheresis Systems) were primarily related to hypotension secondary to changes in extracorporeal volume (>1%)
INDICATIONS
IMPORTANT SAFETY INFORMATION
CONTRAINDICATIONS
The THERAKOS CELLEX Photopheresis System is not designated, sold, or intended for use except as indicated.
Certain underlying medical conditions contraindicate THERAKOS Photopheresis, including patients:
-
who cannot tolerate extracorporeal volume loss during the leukocyte-enrichment phase
-
exhibiting idiosyncratic or hypersensitivity reactions to 8-methoxypsoralen/psoralen compounds
-
with coagulation disorders
-
who have had previous splenectomy
WARNINGS AND PRECAUTIONS
-
THERAKOS Photopheresis treatments should always be performed in locations where standard medical emergency equipment is available. Volume replacement fluids and/or volume expanders should be readily available throughout the procedure
-
Patients who may not be able to tolerate the fluid changes associated with extracorporeal photopheresis should be monitored carefully
-
Procedures, such as renal dialysis, which might cause significant fluid changes (and expose the patient to additional anticoagulation) should not be performed on the same day as extracorporeal photopheresis
-
Individual patients may require a heparin dosage that varies from the recommended dose to prevent post-treatment bleeding or clotting during a treatment
ADVERSE REACTIONS
-
Hypotension may occur during any treatment involving extracorporeal circulation. Closely monitor the patient during the entire treatment for hypotension
-
Transient pyretic reactions, 37.7-38.9°C (100-102°F), have been observed in some patients within six to eight hours of reinfusion of the photoactivated leukocyte-enriched blood. A temporary increase in erythroderma may accompany the pyretic reaction
-
Treatment frequency exceeding labeling recommendations may result in anemia
-
Venous access carries a small risk of infection and pain








